Hollow fiber membrane oxygenators (HFMO), which have been used since the 1950s to provide patients with temporary (up to six hour) oxygen support during a cardiopulmonary bypass (CPB), are today being increasingly used for other therapies, such as acute respiratory distress syndrome (ARDS), and chronic obstructive pulmonary disease (COPB), that can last for up to 20-30 days at a time.
This extended contact time between hollow fiber membranes and blood increases the complications of the natural blood biochemical process of “coagulation”, which diminishes oxygen transfer and increases the pressure drop, ultimately causing the oxygenators to fail. The study described in this article was designed to obtain greater insight into the gas transfer mechanism at microscopic scale using computational fluid dynamics in order to accelerate design exploration to find the optimal solution.
Read the articleCASE STUDY
The text provides an in-depth account of Stefano Odorizzi’s journey in founding and growing EnginSoft, our engineering company specializing in computer simulation and modelling. Established in 1984, EnginSoft overcame early challenges, such as the high cost of computing, to emerge as a leader in simulation services, particularly in the fields of mechanical engineering and computational fluid dynamics (CFD). The narrative highlights several key milestones in the company’s history.
cfd metal-process-simulation industry4 news mechanics optimization
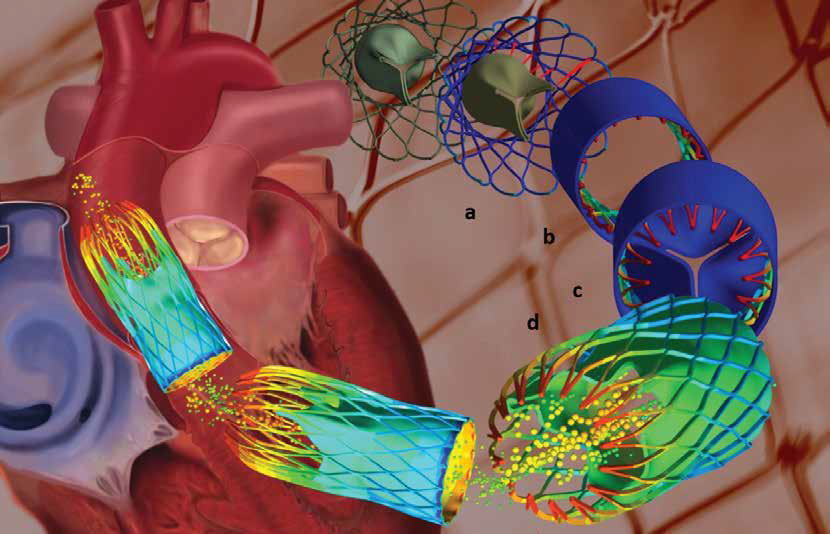
CASE STUDY
Since this cannot be accurately measured in an implanted stent, manufacturers decided to use Multiphysics to simulate the process to better understand the method and to calculate the forces operating on the implant in order to improve the stent design and the surgical procedure, as described in this article.
cfd biomechanics ansys